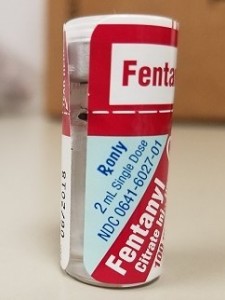
Shared with permission from the April 2017 edition of the ISMP Nurse Advise-ERR®
A pharmacist contacted ISMP to report a diversion and tampering incident that had occurred at his hospital, which was felt to be related to the labeling style of the West-Ward Pharmaceuticals fentaNYL 50 mcg/mL, 2 mL vial. The paper labeling extends to the top of the flip-top cap (Figure 1). This had been carefully pulled back, and the flip-top cap had been removed. Then, the fentaNYL medication had been withdrawn from the vial and replaced with sterile water for injection. The flip-top cap was then replaced and held in place by the label, which had enough adhesive to keep it in place, disguising the tampering and diversion. In fact, the label has enough glue to cause the vials to stick together in the 25-vial carton, sometimes resulting in torn or crinkled labels on vials that have not been tampered with. ISMP contacted West-Ward but has not yet heard whether any changes are planned.
© Institute for Safe Medication Practices Nurse AdviseERR®. FentaNYL diversion alert. April, 2017. Reprinted with permission from ISMP. Sign up for the Nurse AdviseERR® at http://www.ismp.org/newsletters/. Report medication errors or near misses to the ISMP Medication Errors Reporting Program (MERP) at 1-800-FAIL-SAF(E) or online at www.ismp.org.